 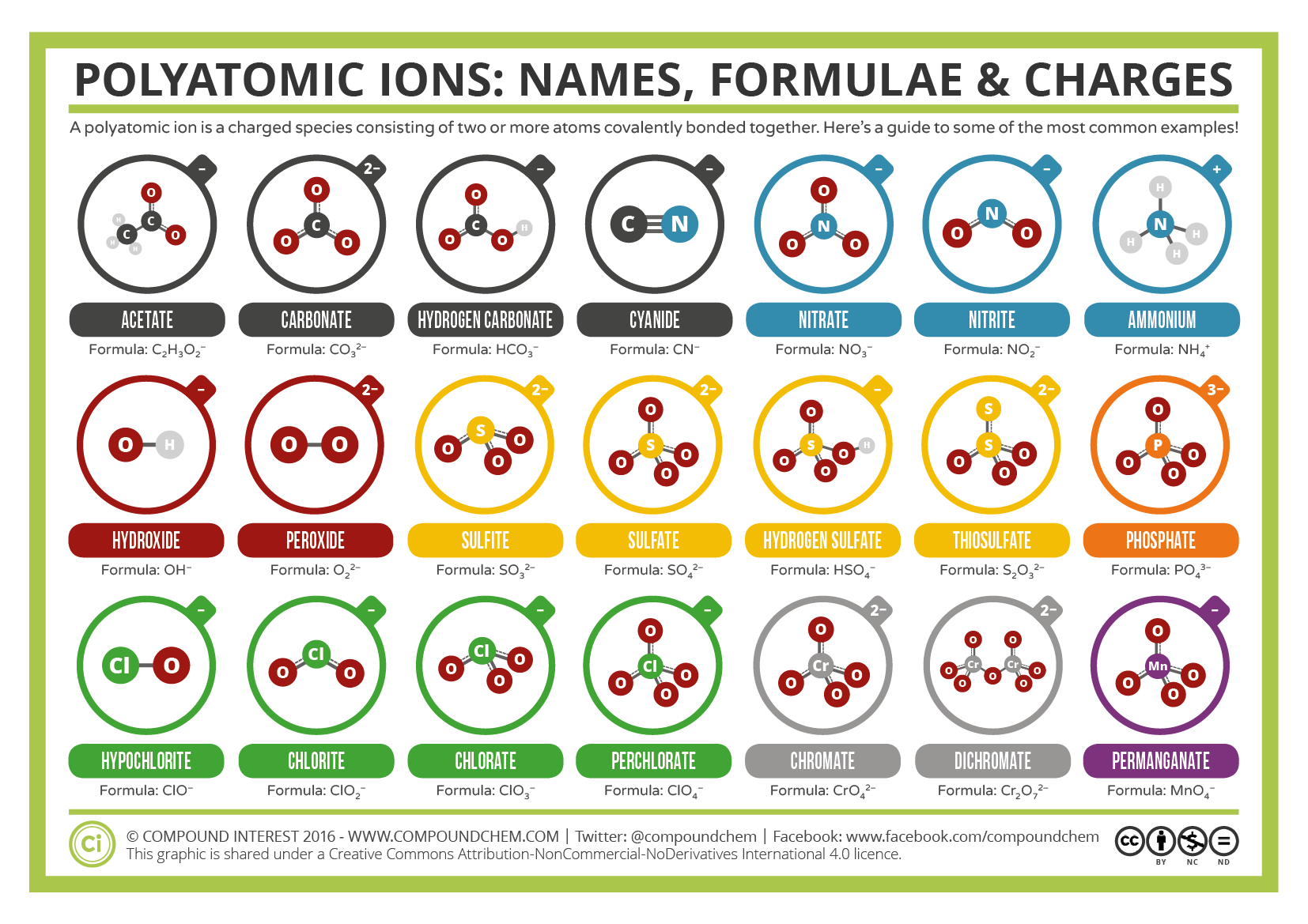
You must determine the relative numbers of ions by balancing the positive and negative charges. Visualize trends, 3D orbitals, isotopes, and mix compounds. There also exists a group of ions that contain more than one atom. Hopefully this helped, you can always add word to the sentence to help remember other polyatomic ions. Interactive periodic table showing names, electrons, and oxidation states. The periodic table can be used to determine which elements are metals and nonmetals. Camel Carbonate 3 consonants 3 O's 2 vowels 2- COX3X2 C O X 3 X 2. To produce a neutral compound, one Sr 2 + is needed for every two NO A 3. Nitrate is a polyatomic ion with formula NO A 3. The compound’s name does not indicate that there are two nitrate ions for every barium ion. The number of vowels stand for the charge. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. Here is a trick to predict the charges on polyatomic ions if you forget them. Strontium is an element in group 2 of the periodic table it forms monatomic Sr 2 + ions. So, this makes the ion have a negative charge.\)? When we gain more electrons, we can know that the amount of electrons is higher than the proton. So, we can conclude that this ion has a positive charge and vice versa. So, the dominant charge of the certain ion is the proton. Practice Problem 6: Name the following ionic compounds. Table 3.3 Common Polyatomic Ions lists the formulas, charges, and names of some common polyatomic ions. Sulfur has a positive oxidation number in SO 2, for example, because it is below oxygen in the periodic table. The periodic table (Figure 3.5 A Simple Periodic Table) can be used to determine which elements are metals and nonmetals. Unlike protons and neutrons, electrons can be easily removed and added to an. Polyatomic ions include (NH 4) +, (CO 3) 2-, and OH. Ions are single charged atoms (simple ions) or small charged molecules (polyatomic ions). If the ion loses the electron, this means that the negative charge is lesser than the positive charge (proton). The sum of the oxidation numbers in a polyatomic ion is equal to the charge on the ion. An ion is a small electrically charged particle. Actually, to understand the ion, we can see how the atoms lose their negative charge (electron). Some of the more important polyatomic ions are listed in. Actually, we have discussed this before above. Note the usefulness of the periodic table in predicting likely ion formation and charge (Figure 2). Examples of monatomic ions include Na +, Fe 3 +, Cl, and many, many others. This differentiates polyatomic ions from monatomic ions, which contain only one atom. Carbonate (CO3) is the polyatomic ion and possesses a charge of -2 By crisscrossing these charges, the ratio of 2:2 reduces to a 1:1 ratio and the resulting formula would have no additional. Whether an atom forms a cation or an anion depends on its position. The examples include cations like ammonium ion ( NH+4 NH 4 + ), and hydronium ion ( H3O+ H 3 O + ) and anions like hydroxide ion ( OH OH ), and. Predicting Cations and Anions based on the Periodic Table. The prefix poly- means many, so a polyatomic ion is an ion that contains more than one atom. Polyatomic ions are molecular ions composed of two or more atoms bonded by covalent bonds and acting as a single unit, but unlike molecules, they have a net charge on them. When we download a periodic table of ions, we also need to know about how to determine an ion. In this article, we will discuss polyatomic ions. Then, if it is reversed, which means the amount of electrons is higher than the proton, we can call it anion. The cation is where the positive charge (proton) has more amount than the electron. There are two kinds of ions in the periodic table of ions. 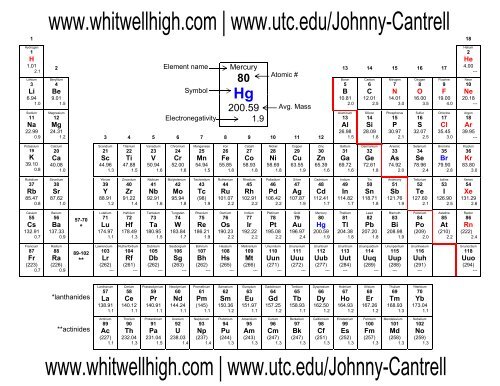
However, it is pretty normal to be found that there is an imbalance of the charge, whether the number of protons is higher or the number of electrons is higher. Well, there are two kinds of ions on the periodic table.Īctually, if an atom or atoms that have equal numbers of negative charge (electrons) and positive charge (proton), then they can be defined as a neutral atom. Yes, one of the things that is required to be understood in the beginning is the ions themselves. 
Yes, we can call this a periodic table of ions.īefore we download the periodic table of irons, we actually need to understand the basic things inside the periodic table of ions. When it comes to learning Chemistry lessons, we may have seen our teacher introduce us with a sheet of the grid that consists of many abbreviations as a symbol of the elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
